Health Canada’s updated Listeria policy has raised expectations for environmental monitoring in ready-to-eat food plants. This guide explains why legacy EMPs fall short, what regulators now expect, and how to redesign a risk-based, data-driven Listeria program that stands up to CFIA and Health Canada scrutiny while supporting stable operations.
When a CFIA inspector arrives without notice, microbiology documentation determines whether the visit stays routine or escalates. This guide explains the microbiology records CFIA expects, common documentation gaps that trigger enforcement, and how to build an audit-ready system that protects your operation, products, and reputation.
Key Takeaways for Executives CFIA looks at your microbiology program as the verification engine of your SFCR Preventive Contro...
Dry foods, such as pistachios, spices, cereals, and milk powders, may appear low risk due to their lack of moisture. But in reality, they can harbor dangerous pathogens like Salmonella, Listeria monocytogenes, and E. coli O157:H7 that can survive for long periods in dry environments (1,2). These organisms don’t need water to persist, and some, like Salmonella, can become more heat-resistant when suspended in low-moisture, high-fat food matrices (3)......
We are pleased to announce that our latest peer-reviewed research article, “Construction and Functional Characterization of a Heterologous Quorum Sensing Circuit in Clostridium sporogenes,” has been officially published in ACS Synthetic Biology....
On Sep 25, 2025, CREM Co Teams, Dr. Zargar, Dr. Rahimifard, and Dr. Lefebre visited the Hans Dairy facility in Mississauga.
Food manufacturers know that a product’s shelf life is critical for quality, safety, and brand reputation. Yet microorganisms – bacteria, yeasts, and molds – can quietly sabotage shelf life, causing spoilage, food safety hazards, and even costly recalls. ...
On August 15, 2025, the Public Health Agency of Canada (PHAC) released an update on the ongoing outbreak of Salmonella ...
On August 15, 2025, the Public Health Agency of Canada (PHAC) released an update on the ongoing outbreak of Salmonella ...
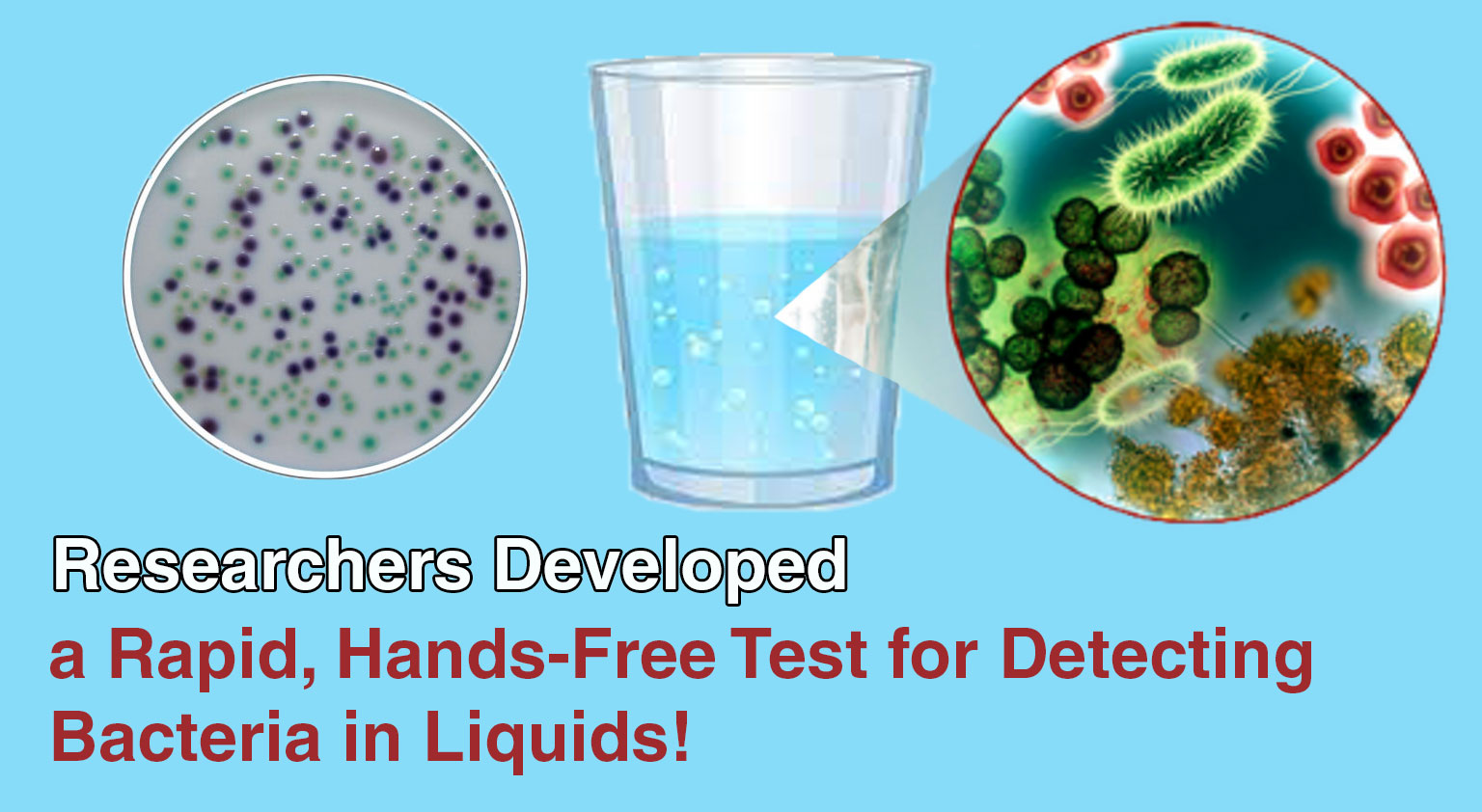
Engineers and biochemists at McMaster University have developed a biogel test that allows untrained users to detect bacterial contamination in ...
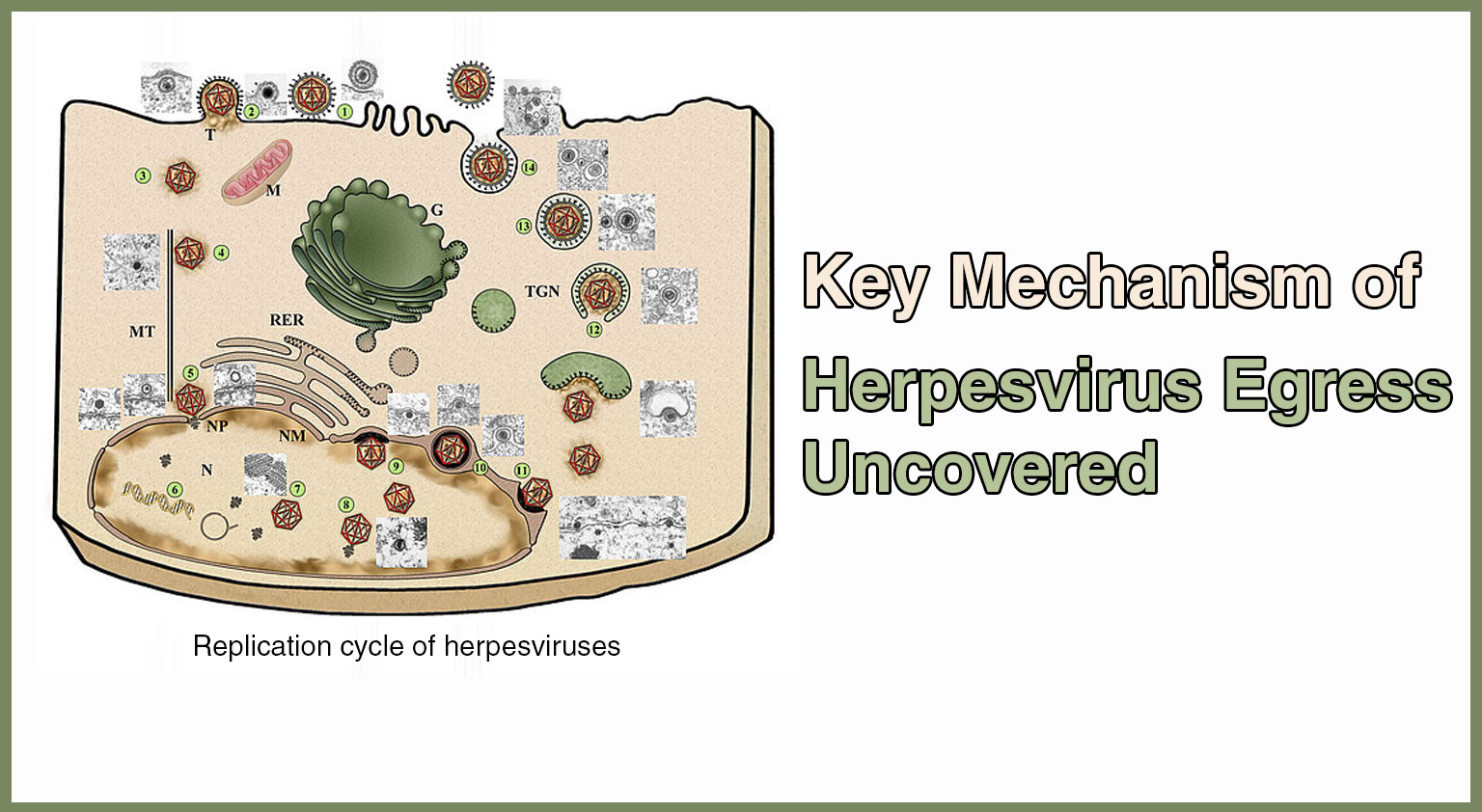
A recent study published in Nature Microbiology on June 25. has shed light on a critical aspect of herpesvirus infections. Herpesviruses are a widespread family of viruses ...
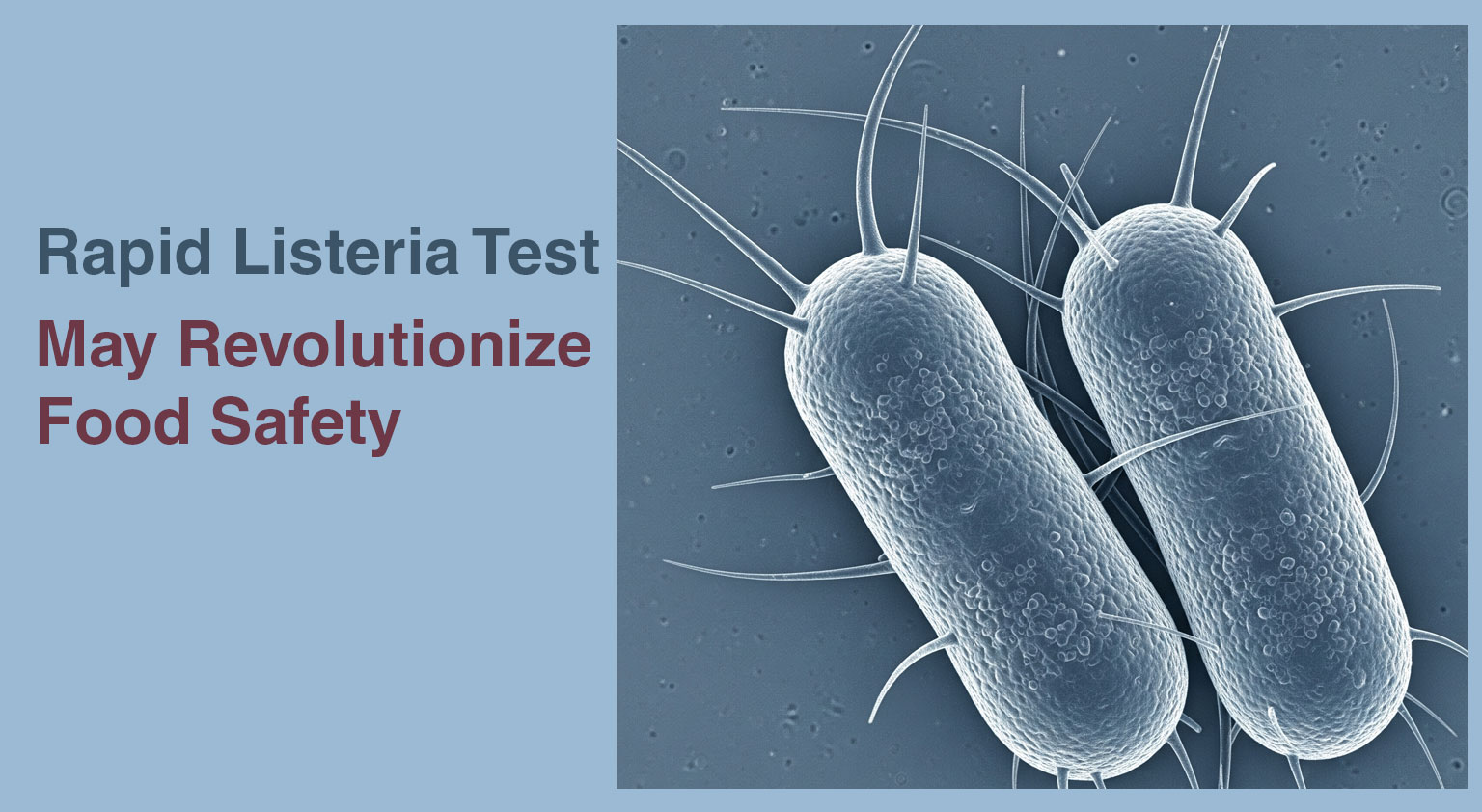
Researchers at Nofima developed a faster method to detect Listeria monocytogenes in food products, which is a significant challenge for the ...
Zebrafish (Danio rerio) larvae have emerged as a powerful vertebrate model in early-stage drug discovery. Approximately 84% of hum...
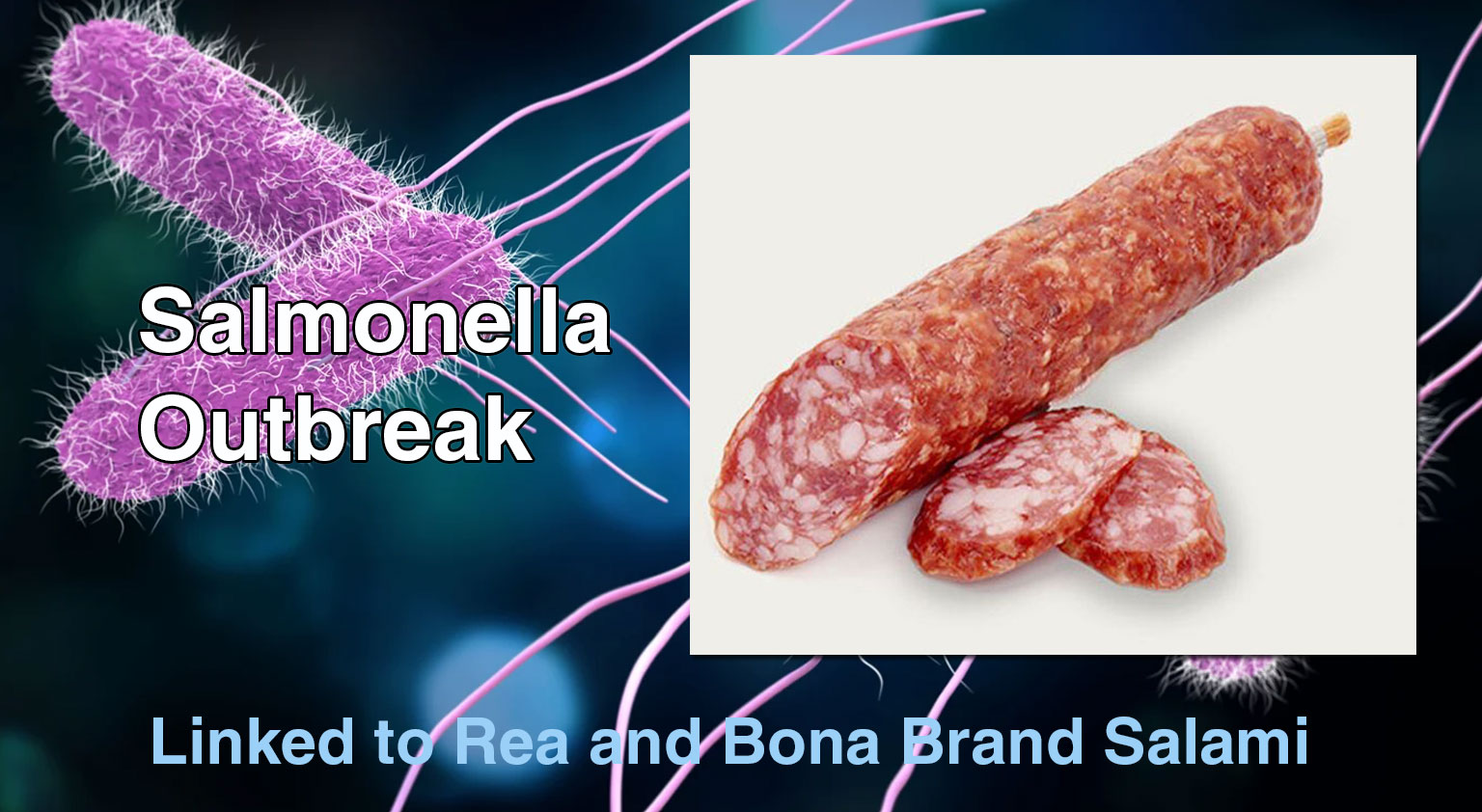
The Public Health Agency of Canada (PHAC) is investigating a multi-province outbreak of Salmonella infections linked to certain Genoa salami products ...
CREM Co Labs is excited to announce its participation in the APIC 2025 Annual Conference & Expo in Phoenix ...
We are pleased to let you know that CREM Co Labs will be exhibiting at IPAC Canada 2025 in Winnipeg, May 30–June 3. We’d love to welcome you at Booth #44 to share Infection Prevention & Control Research, Efficacy and Stability Testing Services, Pathogen ...